Anterior Cervical Plate System | Titanium Cervical Spine Fixation
| Brand | AOYE |
| Certificates | CE/ISO: 9001/ISO13485. Etc |
| Materials | Titanium Alloy |
| MOQ | 1 |
| OEM | Support |
| Lead Time | 7-21Days |
| Logistics | Door to Door,DHL、FedEx、UPS |
- Description
- Advantage
- Application
- FAQ
- Recommended Products
Description
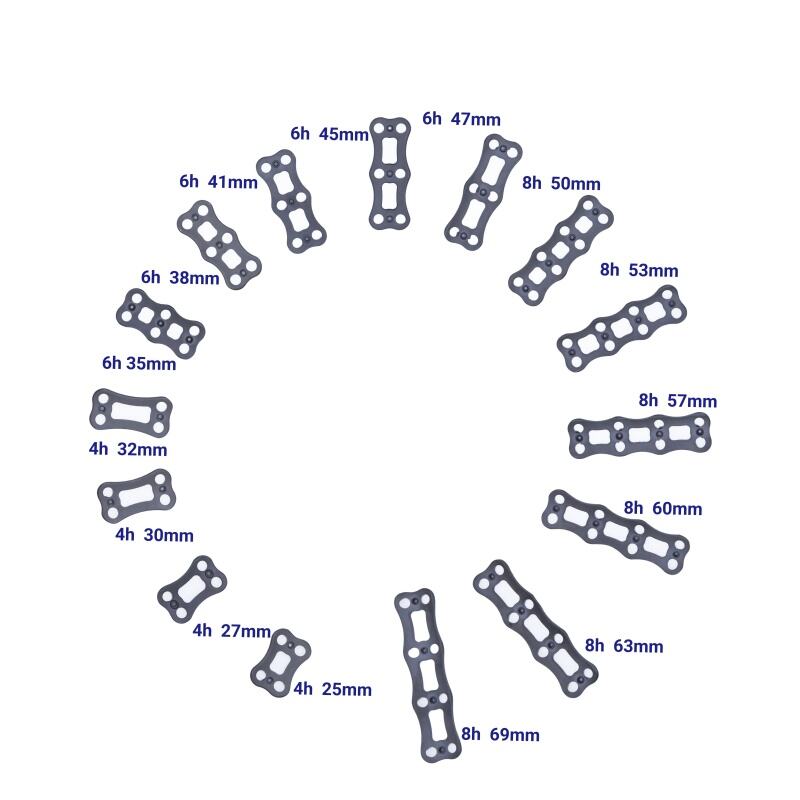
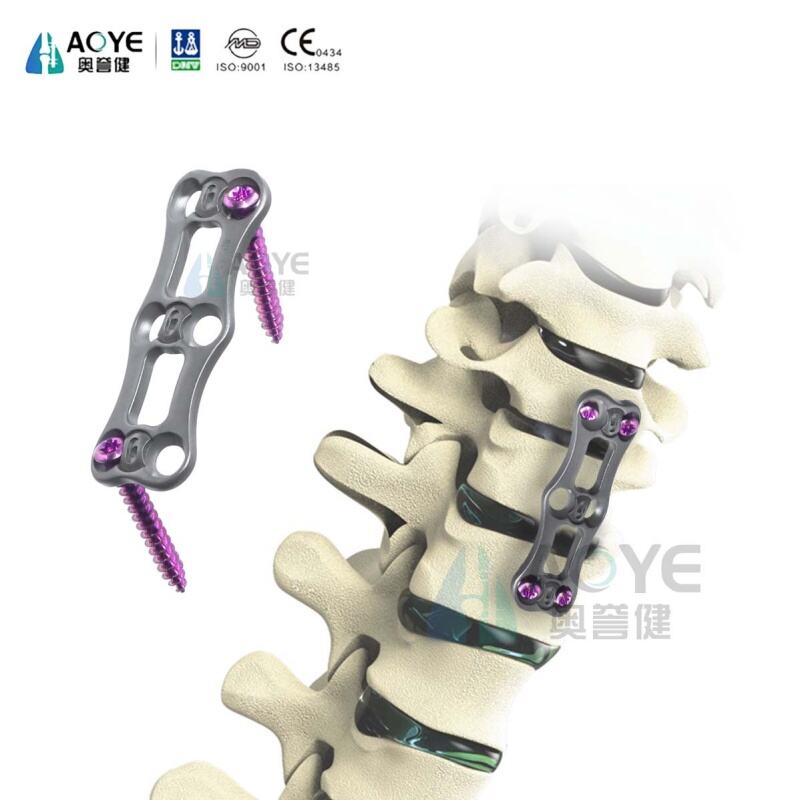
The Anterior Cervical Plate System is designed to provide rigid fixation and stability for cervical spine fusion procedures. Made from high-strength medical-grade titanium alloy, the plate is lightweight, biocompatible, and resistant to corrosion, ensuring long-term durability within the body. Its low-profile anatomical design minimizes irritation to surrounding tissues and reduces the risk of dysphagia after surgery. Multiple screw options, including self-tapping and variable-angle screws, provide versatility for different surgical techniques. The plate features optimized hole positioning for strong fixation while allowing intraoperative flexibility. With streamlined instrumentation and intuitive surgical techniques, the system helps surgeons achieve reliable cervical stabilization and promotes successful bone fusion. CE-certified and ISO13485 compliant, it is trusted by spine surgeons worldwide for anterior cervical procedures.
Advantage
· Anatomical low-profile design to reduce tissue irritation
· Manufactured from medical-grade titanium alloy for strength and safety
· Multiple screw options: fixed-angle and variable-angle
· Streamlined instrumentation for efficient surgical workflow
· Promotes spinal fusion and long-term stability
· CE-certified and ISO13485 compliant manufacturing
Application
· Anterior cervical discectomy and fusion (ACDF) procedures
· Cervical degenerative disc disease requiring stabilization
· Trauma, fractures, and dislocations of the cervical spine
· Revision surgeries requiring additional fixation
· Tumor reconstruction in cervical spine
FAQ
Q1: What is the purpose of an anterior cervical plate?
A1: It provides immediate stabilization of the cervical spine after ACDF or other anterior cervical procedures, supporting bone fusion and preventing implant migration.
Q2: What sizes and lengths are available?
A2: Plates are available in multiple lengths to accommodate 1-level, 2-level, 3-level, and 4-level cervical fusion surgeries.
Q3: What screw types can be used with the plate?
A3: Both fixed-angle and variable-angle self-tapping screws are available to adapt to different patient anatomies and surgical preferences.
Q4: Is the system compatible with standard ACDF cages?
A4: Yes, it is designed to integrate with most commonly used interbody fusion cages.
Q5: What certifications does this product have?
A5: The system is CE-certified and produced under ISO13485 quality standards for medical devices.