Posterior Cervical Screw System | Titanium Cervical Spine Fixation
| Brand | AOYE |
| Certificates | CE/ISO: 9001/ISO13485. Etc |
| Materials | Titanium Alloy |
| MOQ | 1 |
| OEM | Support |
| Lead Time | 7-21Days |
| Logistics | Door to Door,DHL、FedEx、UPS |
- Description
- Advantage
- Application
- FAQ
- Recommended Products
Description
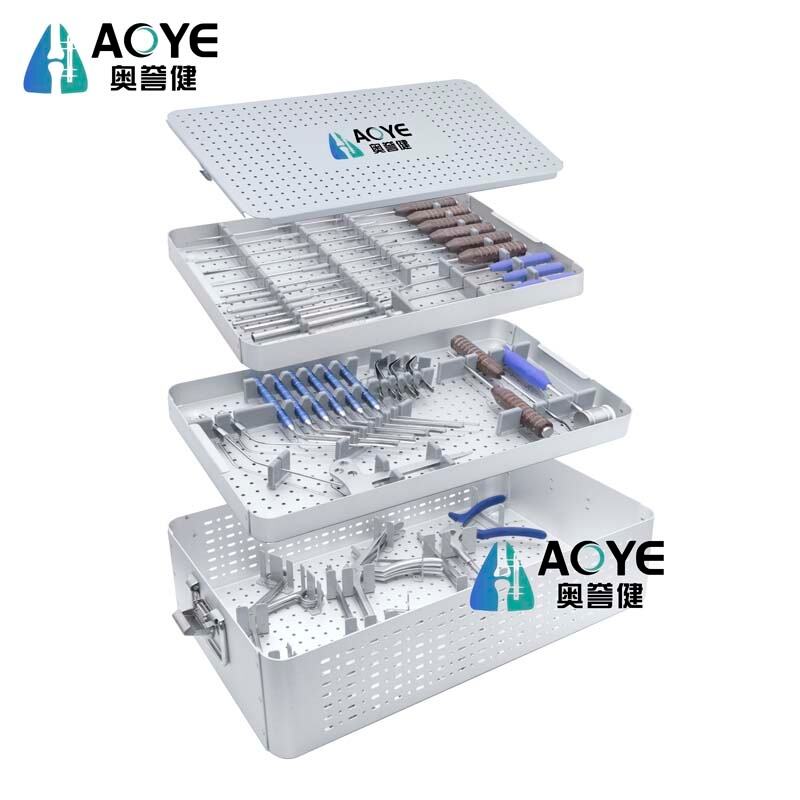
The Posterior Cervical Screw System is engineered to deliver reliable stabilization and fixation in complex cervical spine surgeries. Made from medical-grade titanium alloy, it offers exceptional biocompatibility, strength, and resistance to corrosion. The system includes lateral mass screws, pedicle screws, rods, and cross-links, providing surgeons with a comprehensive solution for posterior cervical stabilization. Its polyaxial screw design allows greater flexibility in rod placement, while self-tapping tips ensure easier insertion and secure anchorage. Low-profile implants minimize soft tissue irritation, and the modular instrumentation is designed for surgical precision and efficiency. This system is CE-certified, ISO13485 compliant, and developed to meet global clinical requirements for treating trauma, deformity, degenerative conditions, and tumor reconstruction in the cervical spine.
Advantage
· Medical-grade titanium alloy for lightweight strength and biocompatibility
· Polyaxial screw design for flexible rod alignment
· Self-tapping screws reduce operative time and bone trauma
· Low-profile implants minimize soft tissue irritation
· Wide selection of screw sizes and lengths for surgical versatility
· CE-certified and ISO13485 compliant manufacturing
Application
· Posterior cervical fixation in trauma, fracture, or dislocation
· Degenerative cervical spine conditions requiring stabilization
· Cervical deformity correction (kyphosis, scoliosis)
· Tumor or infection reconstruction
· Revision surgeries requiring enhanced posterior stability
FAQ
Q1: What is the purpose of an anterior cervical plate?
A1: It provides immediate stabilization of the cervical spine after ACDF or other anterior cervical procedures, supporting bone fusion and preventing implant migration.
Q2: What sizes and lengths are available?
A2: Plates are available in multiple lengths to accommodate 1-level, 2-level, 3-level, and 4-level cervical fusion surgeries.
Q3: What screw types can be used with the plate?
A3: Both fixed-angle and variable-angle self-tapping screws are available to adapt to different patient anatomies and surgical preferences.
Q4: Is the system compatible with standard ACDF cages?
A4: Yes, it is designed to integrate with most commonly used interbody fusion cages.
Q5: What certifications does this product have?
A5: The system is CE-certified and produced under ISO13485 quality standards for medical devices.