Minimally Invasive Pedicle Screw | Titanium Spinal Implant
| Brand | AOYE |
| Certificates | CE/ISO: 9001/ISO13485. Etc |
| Materials | Titanium Alloy |
| MOQ | 1 |
| OEM | Support |
| Lead Time | 7-21Days |
| Logistics | Door to Door,DHL、FedEx、UPS |
- Description
- Advantage
- Application
- FAQ
- Recommended Products
Description
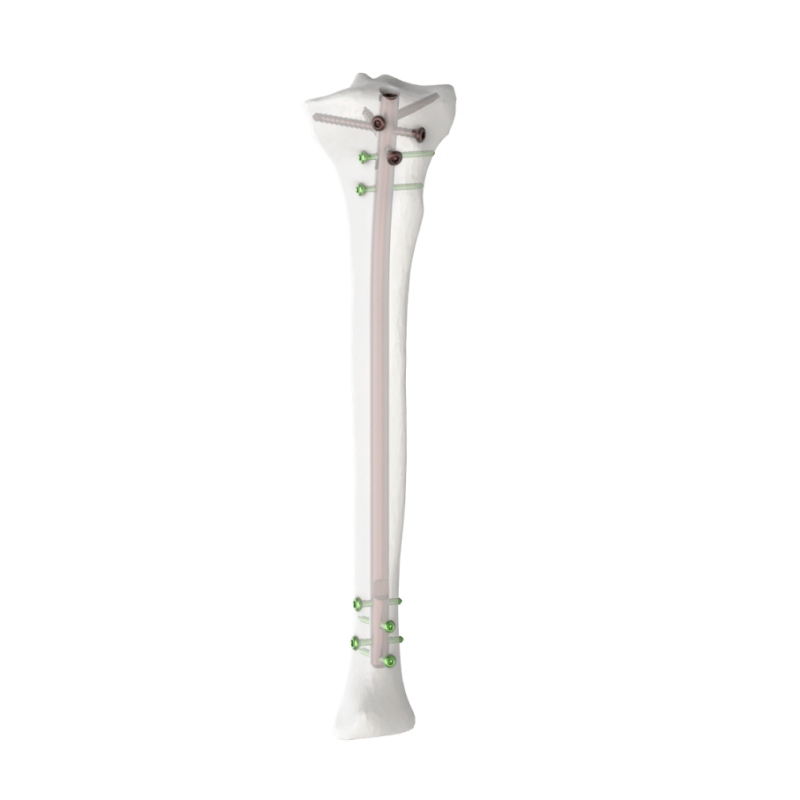
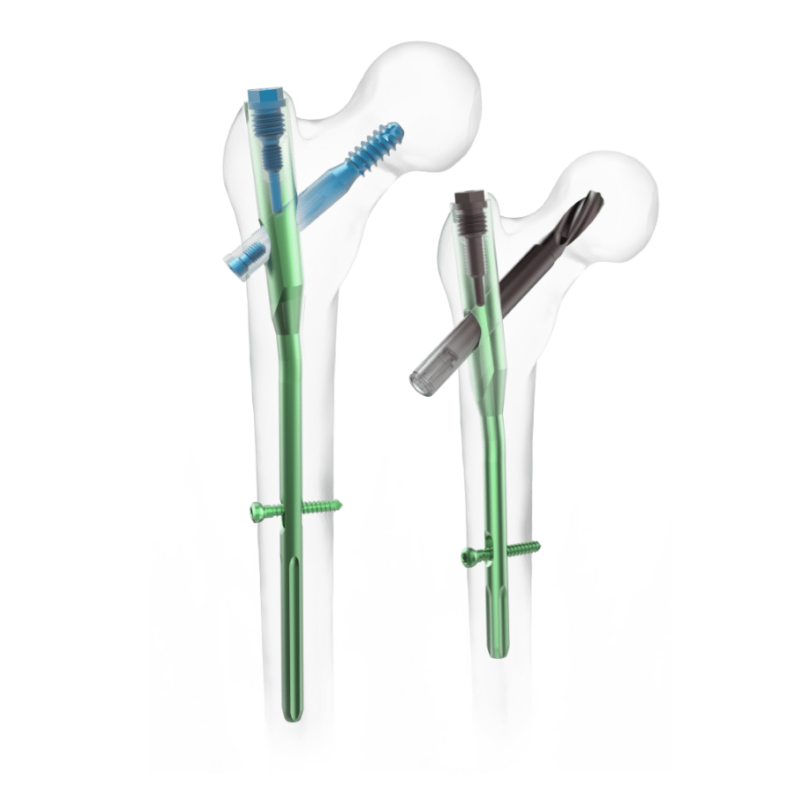
The Minimally Invasive Pedicle Screw is a titanium spinal implant designed for percutaneous fixation in spinal fusion and deformity correction. Manufactured from medical-grade titanium alloy, it offers exceptional strength, corrosion resistance, and biocompatibility. Its polyaxial design and self-tapping thread enable secure anchorage with reduced risk of screw loosening. The low-profile head and optimized thread geometry allow for percutaneous placement through smaller incisions, minimizing muscle disruption and blood loss. Available in various diameters and lengths, the screw system is ideal for minimally invasive spine surgeries, ensuring stable fixation, quicker recovery, and improved surgical outcomes for patients.
Advantage
· Made from medical-grade titanium alloy: strong, lightweight, corrosion-resistant
· Polyaxial head design for versatile rod placement
· Self-tapping thread for reliable bone purchase
· Low-profile head reduces soft tissue irritation
· Available in multiple diameters and lengths
· CE-certified and ISO13485 compliant for B2B procurement
Application
· Minimally invasive spinal fixation procedures
· Percutaneous pedicle screw placement
· Degenerative spine, trauma, and deformity cases
· Spinal fusion and stabilization surgeries
FAQ
Q1: What material is the pedicle screw made from?
A1: It is made from high-quality medical-grade titanium alloy.
Q2: Is this screw suitable for minimally invasive spine surgery?
A2: Yes, it is specifically designed for percutaneous placement in MIS procedures.
Q3: Are different sizes available?
A3: Yes, screws are available in various diameters and lengths for different anatomical needs.
Q4: Can this screw be used in deformity correction cases?
A4: Yes, the polyaxial design and strong fixation make it suitable for trauma and deformity surgeries.
Q5: Who typically purchases this implant?
A5: Spine hospitals, orthopedic departments, and medical distributors specializing in MIS implants.